Hemodynamic Monitor for Ischemic
Stroke Detection
What Are We About?
Problem Statement
In the United States, stroke is the fifth leading cause of death according to data from the Center for Disease Control and Prevention (CDC) [1].
Ischemic strokes are responsible for roughly 87% of these cases. Ischemic stroke occurs when
the vessels responsible for supplying blood to areas of the brain are obstucted. These obstructions cause the neurons in those local regions of the brain to be deprived
of oxygen and nutrients. These localizations are medically termed ischemic penumbra.
These obstructions can be attributed to various conditions involving
thrombotic and embolic clot types. When this occurs, it is critical that the patient be properly diagnosed and treated quickly
as ischemia is a time-sensitive emergency where treatment options are dependent on the temporal stage of the disease.
According to a study published in the American Heart Association journal, it is estimated that 1.9 million
neurons on average are lost every minute a stroke is left untreated. When compared to rates of neuron loss in normal brain aging, this
equates to 3.6 years of accelerated brain aging each hour the ischemic obstruction is left untreated as illustrated in Figure 1 [2]. The longer the obstruction persists, the
probability of permanent neurological damage, poorer clinical outcome, and death increases.
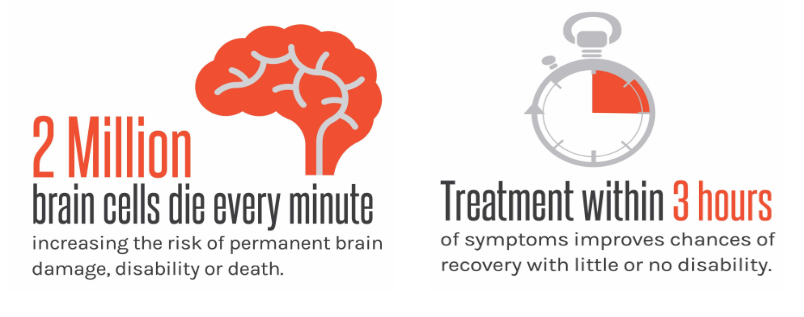
Figure 1: Consequence of leaving ischemic stroke untreated [3]
When a patient is admitted to the critical care units for a potential stroke, current stroke assessment methods adhere to the National
Institute of Health Stroke Scale (NIHSS) testing guidelines to evaluate the severity of the disease [4].
In addition to patient and witness recounts in the field, procedures that are included in these assessments are tests for levels of consciousness, horizontal eye movements
(best gaze), visual field,
motor movements, and speech proficiency [4]. These primary stroke indicators are followed by medical imaging scans such as Magnetic Resonance Imaging (MRI) and Computed
Tomography (CT) scans. These current assessment methods are effective; however, they can consume the critical window of time necessary to employ certain treatment options.
Therefore, ischemic stroke is a complex disease that requires quick and reliable action once debilitating symptoms arise. As mentioned earlier, the rate of neuron loss is significant and
will result in the formation of non-functional scar tissue from necrosis (cell death from injury). Even with prompt hospital assessments, diagnostics, and treatments,
there will always be a probability of long term disability and a significantly reduced quality of life.
Our Goals and Project Aims
Our team has been working hard evaluating the use of a capacitive blood pressure sensor developed from Dr. Michelle Khine's laboratory
in the detection of
ischemic stroke. Our project is broken up into 3 parts:
1) Research and elucidate literature-based evidence surrounding physiological biomarkers like heart rate variability (HRV) as well as blood pressure variability (BPV) and their utility as
a stroke diagnostic.
2) Test and validate capacitance-based pressure sensors for use in continuous blood pressure monitoring through an experimental approach utilizing a hemodynamic
vasculature model and pulsatile pump. This is to
verify if there are statistically significant changes in the blood pressure waveforms from simulated large vessel occclusions.
3) Utilize 3D Computer-Aided Design (CAD) to model an external wearable device to place sensors on patient's carotid arteries.
There is minimal research in using a blood pressure sensor based approach to detecting stroke, let alone a commercially viable method in the market for detecting one.
This technology has the potential to positively impact victims of ischemic stroke and also patients at high risk for one.
This development of a novel detection method to signal the onset of stroke would significantly better equip our health care system against this disease.
References
[1] “Stroke Facts,” Centers for Disease Control and Prevention, 31-Jan-2020. [Online]. Available: https://www.cdc.gov/stroke/facts.htm. [Accessed: 13-May-2020].
[2] J. L. Saver, J. L. Saver, Jeffrey L. Saver From the Stroke Center, J. L. Saver, Stroke Center, Department of Neurology, and Department of Neurology, “Time Is Brain-Quantified,” Stroke, 08-Dec-2005. [Online]. Available: https://www.ahajournals.org/doi/10.1161/01.str.0000196957.55928.ab. [Accessed: 13-May-2020].
[3] D. Tong, D. Tong, M. J. Reeves, M. J. Reeves, A. F. Hernandez, A. F. Hernandez, X. Zhao, X. Zhao, D. W. M. Olson, D. W. M. Olson, G. C. Fonarow, L. H.
Schwamm, E. E. Smith, E. E. Smith, L.
Caplan, California Pacific Medical Center, Cpmc, California Pacific Medical Center, Cpmc, California Pacific Medical Center, Cpmc, California Pacific Medical Center, Cpmc,
California Pacific Medical Center,
Cpmc, California Pacific Medical Center, Cpmc, Lee H. Schwamm From the California Pacific Medical Center, Cpmc, California Pacific Medical Center, and Cpmc, “Times From
Symptom Onset to Hospital Arrival in the
Get With The Guidelines–Stroke Program 2002 to 2009,” Stroke, 26-Apr-2012. [Online].
Available:
https://www.ahajournals.org/doi/full/10.1161/STROKEAHA.111.644963?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub=pubmed. [Accessed: 14-May-2020].
[4] W. J. Powers, William J. Powers Search, and A. A. Rabinstein, “Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association,” Stroke, 30-Oct-2019. [Online]. Available: https://www.ahajournals.org/doi/10.1161/STR.0000000000000211. [Accessed: 14-May-2020].
